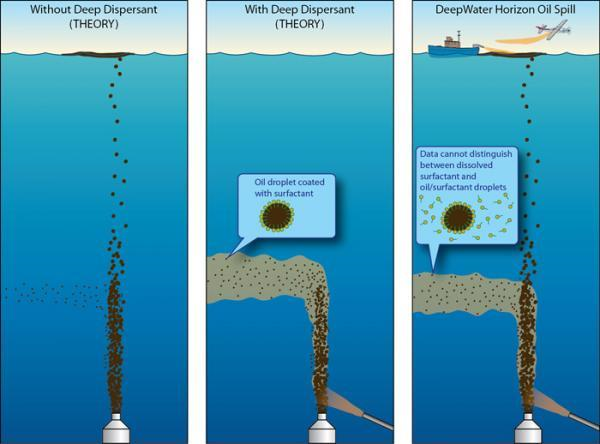
ScienceDaily (Jan. 26, 2011) — To combat last year's Deepwater Horizon oil spill, nearly 800,000 gallons of chemical dispersant were injected directly into the oil and gas flow coming out of the wellhead nearly one mile deep in the Gulf of Mexico. Now, as scientists begin to assess how well the strategy worked at breaking up oil droplets, Woods Hole Oceanographic Institution (WHOI) chemist Elizabeth B. Kujawinski and her colleagues report that a major component of the dispersant itself was contained within an oil-gas-laden plume in the deep ocean and had still not degraded some three months after it was applied.
While the results suggest the dispersant did mingle with the oil and gas flowing from the mile-deep wellhead, they also raise questions about what impact the deep-water residue of oil and dispersant -- which some say has its own toxic effects -- might have had on environment and marine life in the Gulf.
"This study gives our colleagues the first environmental data on the fate of dispersants in the spill," said Kujawinski, who led a team that also included scientists from UC Santa Barbara. "These data will form the basis of toxicity studies and modeling studies that can assess the efficacy and impact of the dispersants.
"We don't know if the dispersant broke up the oil," she added. "We found that it didn't go away, and that was somewhat surprising."
The study, which appears online Jan. 26 in the American Chemical Society (ACS) journal Environmental Science &Technology, is the first peer-reviewed research to be published on the dispersant applied to the Gulf spill and the first data in general on deep application of a dispersant, according to ACS and Kujawinski. Some previous studies had indicated that dispersants applied to surface oil spills can help prevent surface slicks from endangering marshes and coastlines.
Kujawinski and her colleagues found one of the dispersant's key components, called DOSS (dioctyl sodium sulfosuccinate), was present in May and June -- in parts-per-million concentrations--in the plume from the spill more than 3,000 feet deep. The plume carried its mixture of oil, natural gas and dispersant in a southwest direction, and DOSS was detected there at lower (parts-per-billion) concentrations in September.
SEE THE WHOLE STORY HERE:
http://www.desdemonadespair.net/2011/01/first-study-of-dispersants-in-gulf.html
No comments:
Post a Comment